If you have commercial insurance, you may pay as little as $5 for the 12-week course of therapy.
Based on insurance eligibility, restrictions may apply.*
If you have commercial insurance, you may pay as little as $5 for the 12-week course of therapy.
Based on insurance eligibility, restrictions may apply.*
VIVJOA is the first FDA-approved medication for recurrent vaginal yeast infections.
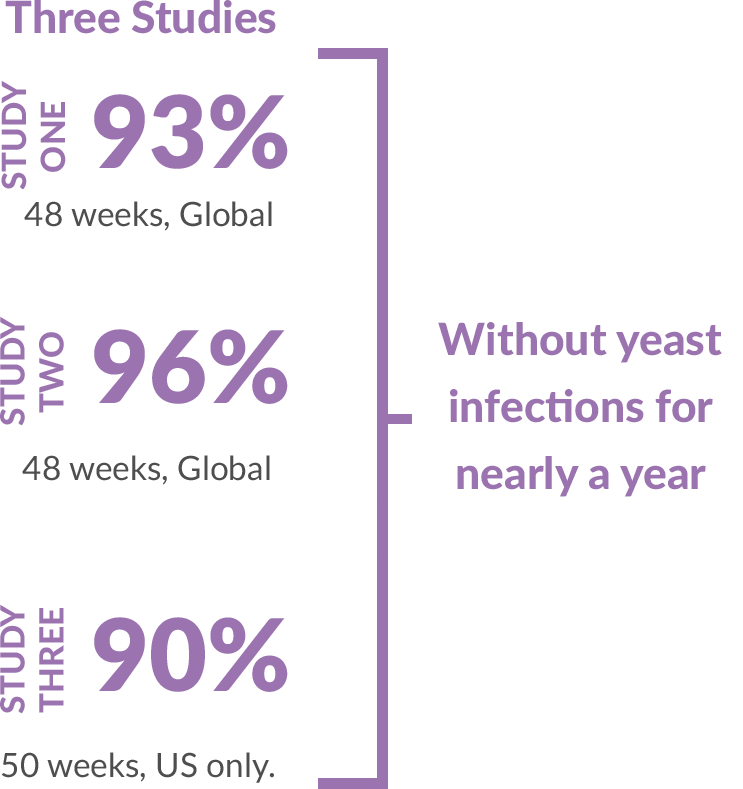
VIVJOA was safe and well-tolerated in all three studies.
The most frequently reported adverse reactions among patients who received VIVJOA included headache (7.4%) and nausea (3.6%).1
Recurrent Yeast Infection, also known as recurrent vulvovaginal candidiasis (RVVC), is a distinct condition2-4 from acute yeast infection and is defined as three or more symptomatic acute episodes of yeast infection in 12 months.5


Your doctor prescribes VIVJOA.

For faster results, please expect a call from Walgreens and be sure to answer or return any calls you may receive from the pharmacy.

Your VIVJOA prescription is shipped next day delivery direct to your door.

VIVJOA® (oteseconazole) is a prescription medicine used to reduce the frequency of vaginal yeast infections that keep coming back (recurrent vulvovaginal candidiasis, RVVC) in females with a history of RVVC who are not pregnant and are not able to become pregnant.
It is not known if VIVJOA is safe and effective in females who have not had their first menstrual period.
Please see full Prescribing Information and Patient Information.
Call your healthcare provider for medical advice about side effects. You may report side effects related to VIVJOA by calling 1-855-299-0637. If you prefer to report these side effects to the FDA either visit www.FDA.gov/medwatch or call 1-800-FDA-1088.
REFERENCES: 1. VIVJOA (oteseconazole). Prescribing Information. Mycovia Pharmaceuticals, Inc.; 4/2022. 2. Jaeger M, Plantinga TS, Joosten LA, Kullberg BJ, Netea MG. Genetic basis for recurrent vulvo-vaginal candidiasis.Curr Infect Dis Rep. 2013;15(2):136-42. 3. Peters BM, Yano J, Noverr MC, Fidel PL Jr, et al. Candida vaginitis: when opportunism knocks, the host responds. PLoS Pathog. 2014;10(4):e1003965. 4. Sobel, JD. Recurrent vulvovaginal candidiasis. Am J Obstet Gynecol. 2016;214(1):15-21. 5. Workowski KA, Bachman LH, Chan PA, et al. Sexually transmitted infections treatment guidelines, 2021. MMWR Recomm Rep. 2021;70(4):1-187 6. Yano J, Sobel JD, Nyirjesy P, et al. Current patient perspectives of vulvovaginal candidiasis: incidence, symptoms, management and post-treatment outcomes.BMC Women’s Health. 2019;19(1):48
GREAT NEWS!
Your online visit is only $49 $29.
Is treatment right for you?
LETS GET STARTED.
By clicking here you will go to a third-party telemedicine provider.